Scientific Achievement
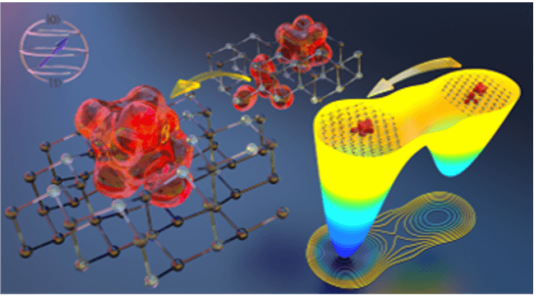
We unraveled the formation mechanism of double vacancies in silicon carbide (SiC), which are promising spin defects for quantum technologies, by using atomistic and quantum simulations.
Significance and Impact
We presented a general simulation strategy coupling advanced sampling techniques and first principles simulations. The strategy can be used to identify novel spin defects in semiconductors and to elucidate their formation mechanism.
Research Details
- We quantified the energetics of divacancy (VV) formation, migration, and reorientation processes in SiC via free energy calculations using density functional theory-based molecular dynamics combined with neural network-based enhanced sampling techniques.
- We found that VV formation is a thermally activated process that competes with the conversion of silicon to carbon vacancy during thermal annealing.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation’s first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America’s scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC for the U.S. Department of Energy’s Office of Science.
The U.S. Department of Energy’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit https://energy.gov/science.